How to Report Drug Side Effects
Reporting drug side effects to the FDA is a key way we can all contribute to ongoing insights into medication side effects and manufacturing problems. Here’s how to submit one
When you start or change medications, MedShadow recommends keeping track of any new symptoms or changes in how you feel so you can recognize potential side effects or shifts in the drug’s effectiveness. It’s important to monitor how you feel over time, since changes in your health (such as weight gain or loss, or a newly diagnosed condition) can affect how well a drug works and whether you experience side effects. Another important change to be alert to is if the manufacturer of your medication has changed. You can sometimes find that name on your prescription label, and if it’s not listed there, you can always ask your pharmacist.
If you notice a change in your medication’s efficacy or develop a new symptom you suspect may be a side effect, your first call should be to your healthcare provider.
We recommend that your next action should be to file a report in the FDA’s MedWatch database.
Why? The Food and Drug Administration (FDA) maintains this system to collect reports of side effects — what it calls “adverse events.” These reports can be submitted by patients, patients’ families, and doctors. The agency regularly reviews these reports to identify trends that may suggest a drug is causing a previously unknown side effect or that there may be problems at a particular manufacturer.
For example, early in the pandemic, reports to the FDA’s Vaccine Adverse Events Reporting System (VAERS) led to a pause in Jannsen’s COVID shot as a potential link to serious side effects was investigated. And in its 2024 Report on the State of Pharmaceutical Quality, the FDA noted an increase in reports of side effects and lack of effectiveness with ADHD stimulants, and is currently evaluating those medications.
Investigations like these can lead the agency to add a new warning to a drug’s label or even remove a drug from the market, but because these reports are not intended to aid any one specific patient, you likely will not hear back from the agency directly. It’s also unclear exactly how often MedWatch reports are monitored.
To report your experience, start by making sure you have as much information as possible, including:
- The name of your medication
- Whether it’s a brand name or a generic drug
- The date you picked up your current refill
- The pharmacy name and address where you purchased it
- When possible, the 10 or 11-digit National Drug Code (NDC) number, the manufacturer, or lot number

You can often find this information on the drug’s label, but your pharmacy can help if it’s missing. If you’ve had blood tests, imaging, or other medical records that support your experience, it can also be helpful to have those on hand so you can enter the data.
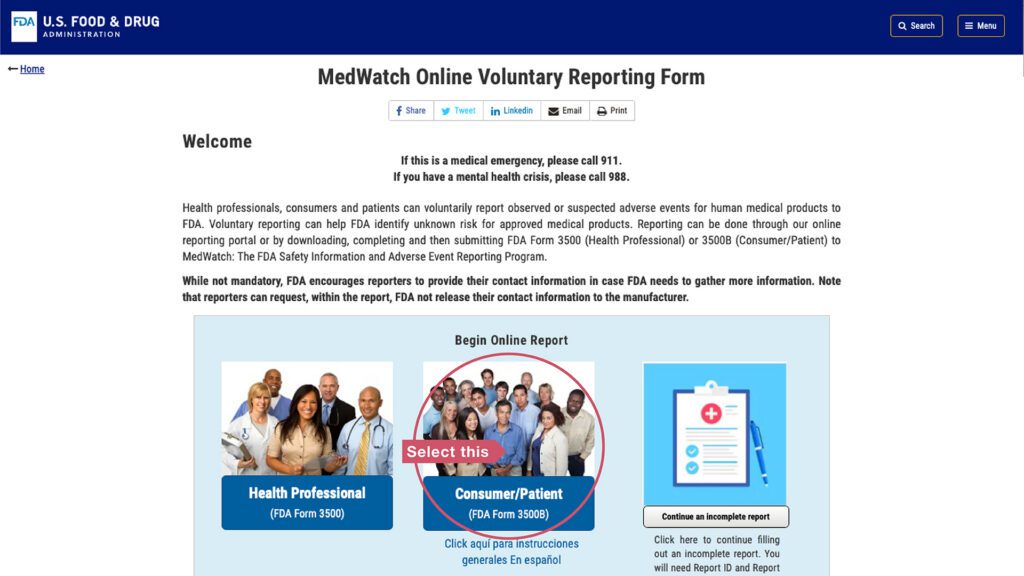
Start on this page. Click the red “report a problem” button.

Then, select the “Consumer/Patient” square. You will be guided through a series of questions to report the problem to the FDA.
If you choose to provide your contact information, the FDA may or may not follow up for more information after you file a report. The person submitting the report must include their first and last name, though the patient can be identified by their initials.

