Press Room
MedShadow’s coverage and commentary highlight our mission to provide science-based information, independent of pharmaceutical influence, to help people to make informed decisions about the RX and OTC medications they take. For media inquiries, contact Lisa Jaycox at press@medshadow.org.
Recent News
Read recent coverage featuring MedShadow’s work and experts.
-
 THE SHORTY AWARDS
THE SHORTY AWARDSShorty Awards: Investigating Generics
MedShadow's Investigating Generics series of articles, webinars, and survey named as a finalist for its creativity and effectiveness.
read more -
 Pharmacoepidemiology & Drug Safety
Pharmacoepidemiology & Drug SafetyCharacteristics of U.S. FDA Drug Recalls Involving Opioid Medications, 2002-2025
This paper analyzes over 20 years of FDA recall data and credits Emma Yasinski and MedShadow for "thoughtful inquiries regarding opioid supply safety that inspired the conception of the manuscript."
read more -
 The Business of Content
The Business of ContentHow a small journalism nonprofit is holding the largest pharma companies to account
MedShadow's Diane Salvatore explains our nonprofit investigative journalism approach, success with social media videos, and what's on the horizon for MedShadow's advocacy and editorial priorities.
read more -
 ADHD Wise Squirrels
ADHD Wise SquirrelsADHD Medication Quality, Oversight, and Reporting
MedShadow's Emma Yasinski explores the hidden complexities of medication safety, the ongoing ADHD medication shortage, and the vital role patients play in drug oversight.
read more -
 United Press International
United Press InternationalCommon painkillers cited as cause of misdiagnosed heart failure
Drugs like gabapentin can cause fluid retention, leading to symptoms that doctors may confuse for heart failure. MedShadow’s article on prescription cascades offers more information.
read more -
 Those Nerdy Girls Blog
Those Nerdy Girls BlogFrom Civil Rights Hubs to Pharmacy Deserts: What Pharmacy Closures Mean—And How You Can Take Action
Pharmacies provide essential care. The loss of a nearby pharmacy hurts the people who rely on it the most. An article in MedShadow’s Meet Me At the Medcart inspired this post.
read more -
 The Street / Yahoo! Finance
The Street / Yahoo! FinanceUS government issues recall for popular blood pressure medicine
Food and drug recalls are a fact of life. In this article, a MedShadow article is cited with an interviewee, Joey Mattingly, quoted about the drug recall process.
read more -
 Live Beyond the Norms Podcast
Live Beyond the Norms PodcastEpisode 118: The Hidden Dangers in Your Medicine Cabinet
MedShadow’s Suzanne Robotti discusses drug trials, ADHD, and the importance of being your own health advocate.
read more -
 The People's Pharmacy
The People's PharmacyWhy Won’t MDs and Pharmacists Believe You When Generic Drugs Fail?
MedShadow’s webinar on Investigating Generic Drugs featured Joe Graedon, David Light, and Katherine Eban.
read more -
 Drug Patent Watch
Drug Patent WatchThe Definitive Guide to Generic Drug Approval in the U.S.: From ANDA to Market Dominance
MedShadow’s Investigating Generics coverage is cited in this comprehensive report on the generic drug market in the U.S.
read more -
 Stat
StatFDA Clinical Trial Inclusivity Guidelines Will Be Back
In this op-ed, MedShadow’s Suzanne Robotti argues that inclusivity guidelines at the FDA are “necessary.”
read more -
 association of healthcare journalism
association of healthcare journalismA Sickle Cell Drug That Was Hyped In Headlines Is Now Suspected Of Harming Patients
MedShadow’s Suzanne Robotti is quoted and advises that neutral language should be used for new drugs, rather than boosterish language like “miracle” or “game-changer.”
read more -
 Stat News
Stat NewsThe FDA should withdraw approval of more than 400 tainted medicines
In this op-ed, MedShadow’s Suzanne Robotti argues that the FDA should have withdrawn 400 drugs from the market after learning they had received fraudulent testing data.
read more -
 National Center for Health Research
National Center for Health ResearchOpen Letter to the FDA: You’re Protecting the Wrong People
An unnamed number of drugs on the market were approved based on potentially fraudulent testing and data. These medications may not be safe or effective, writes Suzanne Robotti.
read more -
 Ask a Patient Health News
Ask a Patient Health NewsTreatments for COPD, excessive sweating, and more
MedShadow Foundation’s article about the DEA’s schedule of drugs is cited in this round-up of drug and treatment news.
read more -
 Better Than a Pill Podcast
Better Than a Pill PodcastEpisode 55: Interview with Suzanne Robotti
Ever wondered who's safeguarding you from medication risks? MedShadow’s Suzanne Robotti speaks about how she turned her own adversity into action.
read more -
 Authority Magazine
Authority MagazineWomen In Wellness: Five Lifestyle Tweaks That Will Help Support People’s Journey Towards Better Wellbeing
From mental health to physical well-being, women like Su Robotti are making significant strides in bringing about change, introducing innovative solutions, and setting new standards.
read more -
 GET SAVVY: DEMYSTIFYING HEALTHCARE
GET SAVVY: DEMYSTIFYING HEALTHCAREEpisode 56: Prescribed and Confused? Ask These 5 Key Medication Questions
Feeling powerless in the face of medication decisions? MedShadow’s Su Robotti's personal journey will surprise and inspire you.
read more -
 Fathers After 50
Fathers After 50Exploring the Hidden Dangers of Prescription Drugs. A Personal Odyssey of Side Effects and Alternative Treatments
Why are we swallowing so many drugs? Are they necessary? Are they safe? MedShadow’s Suzanne Robotti explores lesser-known stories behind medicines.
read more -
 Type 2 Diabetes Talk
Type 2 Diabetes Talk17 Unspoken Truths About Prescription Medication
Discover the untold truths of medication safety with MedShadow’s Suzanne Robotti.
read more -
 Authority Magazine
Authority MagazineBecoming Pain-Free: How to Alleviate Chronic Pain
Pain can lead you to avoid movement, but almost any movement helps lower pain. MedShadow’s Suzanne Robotti shares insights to help those suffering from chronic pain.
read more -
 Med City News
Med City NewsAn Attack on Patients’ Right to Informed Consent
In this op-ed, MedShadow’s Suzanne Robotti discusses claims that Merck failed to warn patients about the risk of an atypical femoral fracture from Fosamax.
read more -
 The Light of Life
The Light of LifeThe Trials and Errors of Prescription Drugs
MedShadow’s Suzanne Robotti shares her personal story and experiences living with the damaging side effects of DES.
read more -
 Veganish and All Things Healthy
Veganish and All Things HealthyEpisode 319: Su Robotti, Founder of MedShadow.org
MedShadow’s Suzanne Robotti discusses MedShadow Foundation’s mission and usefulness to people researching the side effects of medicines.
read more -
 AARP
AARPDES Daughters: A Medication My Mother Took Before I Was Born Will Affect Me for Life
MedShadow’s Suzanne Robotti shares her personal story about a drug that was given to millions of pregnant women with unintended side effects.
read more -
 Politico
PoliticoFDA advisers recommend the agency approve the first over-the-counter birth control pill
As a FDA Advisory Board Consumer member, MedShadow’s Suzanne Robotti weighs in on HRA Pharma’s Opill being approved for sale without a prescription.
read more
News Releases
MedShadow provides regular insights, news and research, and learning opportunities. Read our latest announcements.
-
 Mar 30, 2026
Mar 30, 2026MedShadow’s Investigating Generics Series Selected as a Finalist at the 18th Annual Shorty Awards
In response to demand, MedShadow has improved design, speed, and…
-
 Dec 15, 2025
Dec 15, 2025Redesigned MedShadow.org website provides improved ways to explore side effects and safety of medications
In response to demand, MedShadow has improved design, speed, and…
-
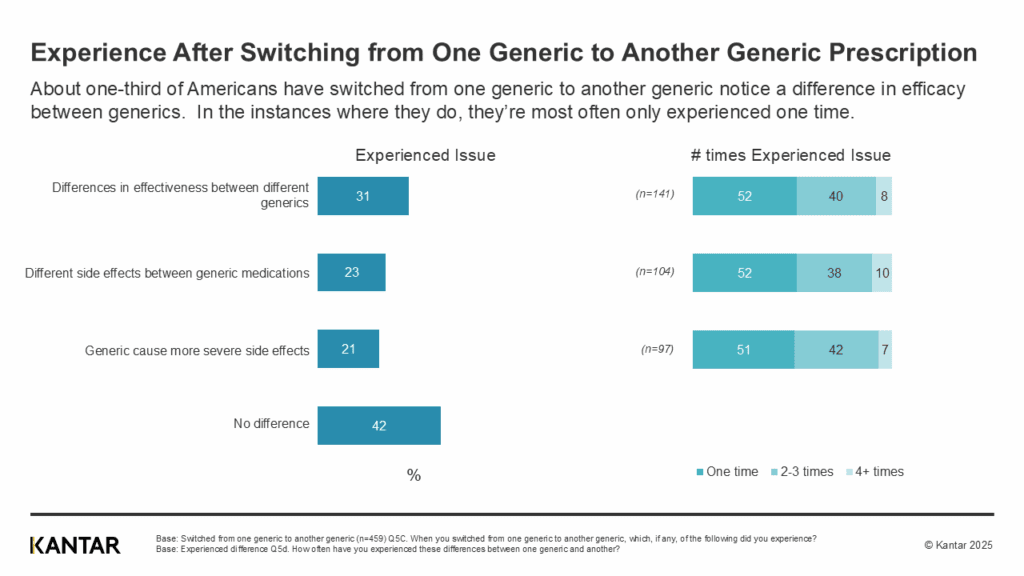 Jul 31, 2025
Jul 31, 2025New Report: Americans Are Willing to Pay More for Drugs Tested for Safety, and Also Report Differences in Side Effects and Efficacy When Switching Between Manufacturers
MedShadow and Kantar's New Data Show Consumer Preferences and Experiences…
-
 May 19, 2023
May 19, 2023MedShadow Founder, President to Speak at PharmedOut Conference
New York, NY - May 19, 2023 - PharmedOut will host…

